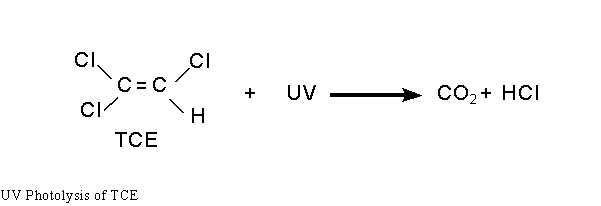
(Photolytic Oxidation Process)
The photolytic oxidation process indirectly destroys volatile organic compounds (VOC) in soil and groundwater. The process uses a xenon pulsed-plasma flash-lamp that emits short wavelength ultraviolet (UV) light at very high intensities. The process strips the contaminants into the vapor phase, and the UV treatment converts the VOCs into less hazardous compounds.
Photolysis occurs when contaminants absorb sufficient UV light energy, transforming electrons to higher energy states and breaking molecular bonds (see figure below). Hydroxyl radicals, however, are not formed. The process requires the UV light source to emit wavelengths in the regions absorbed by the contaminant. An innovative feature of this technology is its ability to shift the UV spectral output to optimize the photolysis.
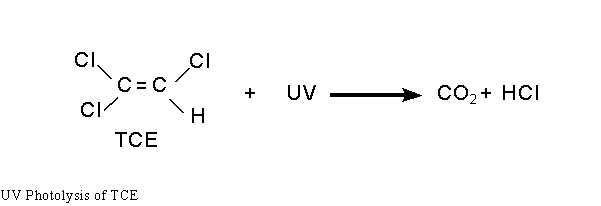
The process uses vacuum extraction or air stripping to volatilize VOCs from soils or groundwater, respectively. VOCs then enter the photolysis reactor, where a xenon flashlamp generates UV light. The plasma is produced by pulse discharge of electrical energy across two electrodes in the lamp. Ninety-nine percent destruction occurs within seconds, allowing continuous operation. Because organics are destroyed in the vapor phase, the process uses less energy than a system treating dissolved organics.
The photolytic oxidation process is designed to destroy VOCs, including dichloroethene (DCE), tetrachloroethene (PCE), trichloroethene (TCE), and vinyl chloride volatilized from soil or groundwater. Destruction of other VOCs, such as benzene, carbon tetrachloride, and 1,1,1-trichloroethane, is under investigation.
The photolytic oxidation process was accepted into the SITE Emerging Technology Program in March 1991. Field testing of a full-scale prototype began in October 1991. The test was conducted at the Lawrence Livermore National Laboratory Superfund site in California. The site contains soil zones highly contaminated with TCE.
During the field test, a vacuum extraction system delivered contaminated air to the unit at air flows up to 500 cubic feet per minute (cfm). Initial TCE concentrations in the air were approximately 250 parts per million by volume. The contaminant removal goal for the treatment was 99 percent. Vapor-phase carbon filters were placed downstream of the unit to satisfy California Air Quality emission control requirements during the field test. Test results are discussed below. The Final Report (EPA/540/R-93/516), the Summary Report (EPA/540/SR-93/516), and the Technology Bulletin (EPA/540/F-93/501) have been published.
The low-wavelength UV emissions allowed direct photolysis of many VOCs, particularly chlorinated compounds and freons, that would not have been possible with commercial mercury vapor lamps. TCE, PCE, and DCE were quickly destroyed. To be rapidly photolyzed, some VOCs require photosensitization or an even lower-wavelength light source.
TCE results are shown in the table below. TCE removal yielded undesirable intermediates.
| TCE PHOTOLYSIS FIELD TEST RESULTS | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Freq. (Hz) | No. of Chambers | Flow (cfm) | Res. Time (sec) | TCE Input (ppmv) | TCE Output (ppmv) | TCE Destruction (%) | DCC Yield (ppmv) | DCAC Yield (ppmv) | Chlorine Balance (Mole%) |
| 30 | 4 | 103 | 9.6 | 78.4 | dl | >99.99 | nd | 20.2 | 78.8 |
| 30 | 4 | 97 | 10.1 | 108.5 | dl | >99.99 | 21.3 | 26.5 | 106.2 |
| 30 | 4 | 95 | 10.4 | 98.3 | dl | >99.99 | 25.6 | 34 | 114.5 |
| 30 | 2 | 106 | 4.6 | 91.7 | 0.07 | 99.92 | 15.9 | 49.2 | 91.1 |
| 15 | 4 | 97 | 10.1 | 106.8 | dl | >99.99 | 22.8 | nd | nd |
| 15 | 2 | 103 | 4.8 | 101.3 | dl | >99.99 | 12.6 | 65.3 | 86.2 |
| 5 | 4 | 95 | 10.4 | 104.9 | dl | >99.99 | 8.7 | 75.7 | 90.0 |
| 5 | 2 | 103 | 4.8 | 101.4 | dl | >99.9 | 9.4 | 76.3 | 88.8 |
| 1 | 4 | 106 | 9.3 | 101.7 | 0.85 | 99.16 | 12.5 | 83.2 | 90.3 |
| 1 | 2 | 103 | 4.8 | 98.5 | 13.23 | 86.57 | 6.8 | 84.9 | 93.3 |
| Notes: Hz = Hertz | |||||||||
| cfm = cubic feet per minute | |||||||||
| sec = seconds | |||||||||
| ppmv = parts per million volume | |||||||||
| dl = detection limit | |||||||||
| nd = not detected | |||||||||
Greater than 85 percent of the TCE chain photo-oxidation product is dichloroacetyl chloride (DCAC). Further oxidation of DCAC is about 100 times slower than TCE photolysis and forms dichlorocarbonyl (DCC) at about 20 percent yield. At this treatment level, the DCC concentration may be excessive, requiring additional treatment.
Further studies should focus on (1) the effectiveness of dry or wet scrubbers for removing acidic photo-oxidation products, (2) development of thermal or other methods for posttreatment of products such as DCAC, and (3) the use of shorter-wavelength UV lamps or catalysts to treat a broader range of VOCs.
FOR FURTHER INFORMATION:
EPA PROJECT MANAGER:
Norma Lewis
U.S. EPA
National Risk Management Research Laboratory
26 West Martin Luther King Drive
Cincinnati, OH 45268
513-569-7665
Fax: 513-569-7787
TECHNOLOGY DEVELOPER CONTACT:
Steve McAdams
Thermatrix, Inc.
101 Metro Drive, Suite 248
San Jose, CA 95110
408-453-0490
Fax: 408-453-0492