|
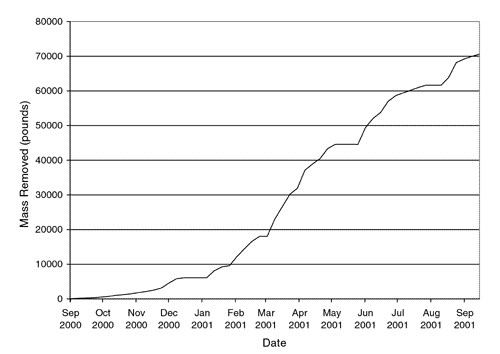
Ground Water Currents, December 2001, Issue No. 42ContentsDUS Expedites Ground-Water Cleanup at Savannah River Site Sodium Dithionite Injections Used for Chromium Reduction Cross-Hole Radar Method Monitors PRB Installation DUS Expedites Ground-Water Cleanup at Savannah River Siteby Michelle Ewart, U.S. Department of Energy, and James Kupar, Bechtel Savannah River, Inc. In September 2000, the U.S. Department of Energy (DOE) began operating a dynamic underground stripping (DUS) system to remove volatile organic compounds (VOCs) at DOE’s Savannah River Site (SRS). At its peak operation, dense non-aqueous phase liquid (DNAPL) was removed at a rate of 1,200 pounds each day. It is estimated that DUS extracted material 15 times faster than the original soil vapor extraction methods and 75 times faster than the pump and treat configuration already operating within the SRS “M-Area.” The M-Area includes an accumulation of several unknown releases over the past 40 years from above-ground tanks used to store solvents. Within this 10,000 square-foot area, DNAPL containing tetrachloroethylene (PCE) and trichloroethylene (TCE) extends to a depth of 165 feet. Typical of the Atlantic coastal plain, the geology in this area consists of clay-rich confining intervals within more transmissive, sandier intervals. The local aquifer is located at a depth of 130 feet. The DUS process used at the M-Area involved a series of steam injections followed by a combined air/steam injection, i.e., hydrous pyrolysis oxidation (HPO). Using electrical resistance tomography in combination with thermocouples, the system’s heating fronts were imaged to monitor progress and schedule the frequencies of injections. DUS does not require the precise identification of contaminant location. The 100- by 100-foot treatment zone at the M-Area extended from a depth of 20 feet below ground surface downward through 110 feet of vadose zone into the water table until reaching the top of the “green clay” confining layer at a depth of 165 feet. Continuous injections applied steam at a temperature of 230°C and maximum rate of 20,000 pounds per hour. Injection wellhead pressures ranged from 40 to 70 pounds per square inch, depending on the lithology of screen intervals, depths, and operational specifications of the system. Two months after initiation of the steaming and extraction process, steam breakthrough occurred at the central extraction well. The HPO process began one month later, and the applied boiling point (90°C) was reached throughout the soil block after another three months. At that point, a total of 45 x 109 British thermal units of steam had been injected. Over the 12 months of active DUS operation, a total of approximately 70,000 pounds of PCE and TCE was removed (Figure 1).
At the peak of operation, vapors from the system’s extraction wells reached a level of 6,600 parts per million volume (ppmv). A total of 3 billion standard cubic feet of air at temperatures approaching 200°F was extracted and cooled over the course of operations. The resulting condensate was directed at a rate of 20 gallons per minute to a surface-level separator where the DNAPL (totaling less than five gallons) was removed from water and dispositioned at an onsite waste facility. Almost 160 million standard cubic feet of PCE- and TCE-bearing air was extracted as noncondensable flow and discharged through the attached soil vacuum extraction unit. Factors found to affect the success of DUS application at the M-Area included the: (1) different injection methods required for varying lithologies, (2) site-specific criteria used to determine optimal shut-down points and processes, and (3) inherent difficulties associated with precise measurement of HPO efficiency. Most significantly, product removal rates were found to be limited by the maximum temperature that could be achieved with this closed DUS system. Following a cool-down period of six months, the system will be further examined to measure the completeness of remediation in the target zone. Total costs for this project are estimated at $4.8 million. DUS operations at the M-Area were designed and conducted by Integrated Water Resources, Inc. (based in Santa Barbara, CA) in cooperation with IT Corporation. In addition, technical assistance for the project was provided by Lawrence Livermore National Laboratory through the DOE’s Subsurface Contaminants Focus Area (SCFA) Lead Laboratory Support Program. A final project report will be available in early 2002 on the SCFA Web site, www.envnet.org/scfa. For more information, contact Michelle Ewart (DOE) at 803-725-1115 or michelle.ewart@srs.gov, or Jim Kupar (Bechtel Savannah River, Inc.) at 803-952-6525 or james.kupar@srs.gov. Sodium Dithionite Injections Used for Chromium Reductionby Cynthia J. Paul, U.S. EPA/Office of Research and Development/National Risk Management Research Laboratory A field-scale pilot study was conducted in 1999 at the U.S. Coast Guard Support Center in Elizabeth City, NC, to evaluate the effectiveness of injecting sodium dithionite into the upper aquifer and lower vadose zone to create a permeable reactive barrier (PRB) system utilizing naturally occurring iron for hexavalent chromium (Cr) (VI) reduction. Within three days of chemical injection, Cr(VI) concentrations dropped to below the target cleanup levels. Based on these results, a full-scale treatment system employing sodium dithionite injections was implemented earlier this year. Past operations of a chrome-plating shop at the Support Center created a contaminant plume with mixed organic and metal compounds, particularly Cr(VI). Due to indications that chromium in the soils would remain immobile and present no potential risk, initial cleanup efforts focused on treating the diffuse chromium plume. A PRB using zero-valent iron was installed at the site in 1996 and has since proved effective for remediating the mobile chromium plume. A water-main break in the immediate vicinity of the plating shop during 1994, however, caused an artificial rise in the water table directly beneath the shop. As a result, soluble Cr(VI) in concentrations reaching 28.0 mg/L had migrated into the ground water. Although concentrations decreased to approximately 4.4 mg/L by 1997, additional treatment was required to meet the maximum contaminant level of 0.1 mg/L for ground water. The U.S. Coast Guard estimated that additional contaminant release into the mobile plume being treated by the PRB would result in the potential need for replacement of the PRB in approximately 10 years. Based on these findings, immediate treatment of the contaminant source was found more cost effective than eventual replacement of the PRB. Prior to the source control pilot study, steps were taken to re-characterize the site through the collection of soil cores from 32 locations within the plating shop. Preliminary screening was conducted using x-ray fluorescence and inductively coupled plasma spectroscopy to determine total chromium levels. Laboratory batch extractions were conducted to selectively remove Cr(VI) from contaminated soils in order to delineate the extent of the soluble, easily mobilized Cr(VI). Results indicated that the contaminant plume had migrated vertically through a series of sand and silt/clay loam layers into the ground water at 2 meters below ground surface and horizontally toward the Pasquotank River, which is located approximately 60 meters from the source area. These conditions provided a continual release of Cr(VI) into the mobile contaminant plume treated by the PRB. Additional laboratory studies were conducted to better understand the potential for in situ reduction/oxidation (redox) manipulation (ISRM) and to evaluate sodium dithionite (Na2S2O4), l-ascorbic acid (C6H8O6), and free hydroxylamine (FH-50™) as potential reductants. Results showed sodium dithionite to be the most effective at reducing chromium with the least adverse side effects. Injecting sodium dithionite into areas with high levels of naturally occurring iron (Fe) was found to create a spatially-fixed reducing zone analagous to a chemical PRB. Through such injection, Fe(III) is reduced to Fe(II) and redox-sensitive contaminants such as Cr(VI) are immobilized or precipitated as they migrate through the reducing zone. In the field pilot, a solution of sodium dithionite (17 kg), potassium bicarbonate (19 kg), and bromide tracer (5.63 kg) in 1,874 liters of distilled and deionized water was injected into ground water at depths ranging from 1.2 to 2.4 meters below ground surface (Figure 2). The reductant was injected continuously for 38 hours at 1 liter/minute. To evaluate the use of ISRM in the capillary fringe areas, test data were collected from the 1-meter treatment area immediately surrounding the injection well.
Field results indicated that Cr(VI) concentrations dropped from pre-treatment levels of 2.2-5.0 mg/L to non-detect levels within 60 hours of injection and remained at virtually the same level throughout the remaining course of the pilot project’s 48-week monitoring period. Data showed that byproducts of the redox process (sulfate and dissolved iron) significantly increased during the initial 60-hour period but decreased to original levels approximately 32 weeks after the injection phase. Similarly, other process indicators such as pH and specific conductance initially rose but returned to or near original levels within approximately 48 weeks, which is typical of the redox process. Pumping and treatment of the injected material was not required because sodium dithionite is known to produce no adverse side effects or byproducts. Costs for the pilot project totaled approximately $750,000, including expenses for soil excavation in the upper area of the treatment zone. Comparative analysis estimated that the addition of chemical injections for source control at the Support Center site helped to reduce the overall project (including mobile plume treatment through the PRB) costs by 40 percent and the cleanup time by 28 years. In comparison to conventional pump and treat methods, analysis estimated that this combined approach for source control and plume treatment achieved nearly an 80 percent reduction in costs. More information on the pilot study is available on the Internet at www.epa.gov/ORD/NRMRL or from Cynthia Paul (National Risk Management Research Laboratory) at 580- 436-8556 or e-mail paul.cindy@epa.gov. Cross-Hole Radar Method Monitors PRB Installationby Jennifer G. Savoie, John W. Lane, Jr., and Peter K. Joesten, U.S. Geological Survey The U.S. Geological Survey (USGS), in cooperation with the U.S. Air Force Center for Environmental Excellence (AFCEE), conducted a study using cross-hole, common-depth radar scanning to determine the lateral and vertical extent of permeable reactive barriers (PRBs) installed near the “Chemical Spill-10” (CS-10) source area at the Massachusetts Military Reservation (MMR) on Cape Cod, MA. The study also enabled researchers to determine whether any large holes in the barriers existed that could allow contaminated ground water to pass untreated through the remediation zone. Based on the survey results, researchers were able to determine that the PRBs were installed in the planned locations. Ground water beneath and downgradient from the CS-10 source area has been affected by contaminants such as chlorinated solvents during decades of past military operations. These contaminants entered the ground-water system and continue to move with the ground-water flow in the underlying sand and gravel aquifer toward nearby streams, ponds, and coastal bays. Depth to ground water at the CS-10 source area is about 80 feet below ground surface (bgs), and the ground-water flow rate is estimated at 0.5-1 foot per day. The contaminant of greatest concern is tetrachloroethene, with concentrations at the source area of approximately 250 micrograms per liter. The University of Waterloo in Ontario, Canada, conducted a field trial involving the installation of two demonstration-width, full-depth, granular-iron walls in the path of the ground-water plume. The depth of the plume (80-120 feet bgs) exceeded practical limits for conventional emplacement methods such as trenching. To address this limitation, hydraulic fracturing and injection methods were used to install the walls. In the field, radar surveys were conducted before and after the iron wall installations. Cross-hole radar scans were conducted in 20 boreholes on opposite sides of the walls using a radar system containing electric-dipole antennas with center frequencies of 100 and 250 Megahertz. Radar-scan measurements of the regions between pairs of boreholes were acquired by moving a radar transmitter and receiver in unison at 10-centimeter increments from the tops of casings to the bottoms of the boreholes. As anticipated, the amplitudes of radar waves crossing the iron zones decreased significantly between the pre- and post-installation surveys, indicating the presence of iron. The results of two-dimensional, finite-difference/time-domain numerical modeling based on laboratory-scale physical models were used to interpret changes in radar amplitude observed in the field. The numerical and physical models simulated a wall of perfectly conductive material embedded in saturated sand that was representative of the MMR field conditions. Modeling results indicated that the amplitude of a radar pulse transmitted across the edge of the conductive wall was about 43 percent of one transmitted through background material. Figure 3 shows the transverse sections for the locations of the two iron walls based on radar scanning results. These data indicate that the “A-Wall” contains two zones: the upper zone is about 33 feet wide, extending 82-102 feet bgs; the lower zone is about 26 feet wide, extending 103-113 feet bgs. The “B-Wall” is interpreted as a 30 foot-wide continuous zone extending about 89-113 feet bgs. It was determined that no holes existed within the boundaries of either iron wall.
The USGS has since employed this radar method at other sites, including the USGS Fractured Rock Hydrology Test Site at Mirror Lake, NH, to locate and characterize bedrock fractures and lithologic changes and to monitor tracer tests. For more information on the use of borehole radar technology at the MMR site, visit the USGS Web site at http://water.usgs.gov/ogw/bgas/publications/wri004145/index.html or contact John W. Lane, Jr., (USGS) at jwlane@usgs.gov. Information on the MMR PRB field trial is available from Rose Forbes (AFCEE) at rose.forbes@mmr.brooks.af.mil or David W. Hubble (University of Waterloo) at dwhubble@uwaterloo.ca.
|