Dioxins
Overview
- Dioxin and Related Compounds
U.S. EPA Web page. - Dioxins and PCBs
U.S. Food & Drug Administration Web page.
Dioxins, or polychlorinated dibenzodioxins (PCDDs), are a group of 75 congeners that differ in the number and location of chlorine atoms. PCDDs are formed as products of incomplete combustion and as byproducts of manufacturing. Although some PCDDs in the environment are released during forest fires and thus natural in origin, they are also released in combustion emissions from municipal and industrial waste incinerators and from burning fossil fuels and wood. As byproducts, PCDDs may be released during the manufacture of chlorinated chemicals (e.g., wood preservatives and herbicides), secondary metal smelting (particularly aluminum, copper, and lead), cement kilns, and chlorine bleaching of wood pulp for paper. PCDDs are not intentionally manufactured, except for use in environmental research.
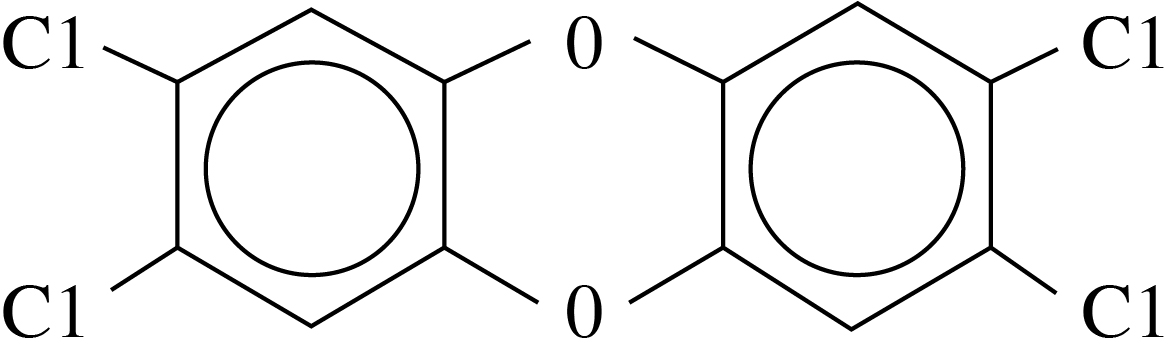
PCDDs are organic compounds, chemically classified as halogenated aromatic hydrocarbons. They are structurally composed of two benzene rings joined by oxygen atoms at the "meta" and "para" positions. The 75 congeners include two monochlorodibenzo-p-dioxins (MCDD), 10 dichlorodibenzo-p-dioxins (DCDD), 14 trichlorodibenzo-p-dioxins (TrCDD), 22 tetrachlorodibenzo-p-dioxins (TCDD), 14 pentachlorodibenzo-p-dioxins (PeCDD), 10 hexachlorodibenzo-p-dioxins (HxCDD), 2 heptachlorodibenzo-p-dioxins (HpCDD), and a single octachlorodibenzo-p-dioxin (OCDD). While this contaminant focus area specifically addresses PCDDs, it is important to note that structurally similar compounds such as chlorinated dibenzofurans (PCDFs) and polychlorinated biphenyl (PCB) congeners are often found with PCDDs. Several of the PCDF and PCB congeners have similar toxic properties and are referred to as "dioxin-like" chemicals with dioxin-like effects.
The behavior of PCDDs in the environment is explained by their physical-chemical properties. PCDDs have relatively low vapor pressures and high octanol/water partition coefficient (Kow) values, which means they do not readily evaporate and tend to sorb onto soil and sediment. The high Kow values also mean that PCDDs will move easily into the fatty tissues of grazing animals when they incidentally ingest them soil adhering to grass. PCDDs bioaccumulate and biomagnify in animal fat. Similarly, when humans consume these animals, the dioxins then accumulate in human fatty tissues. Limited exposure to dioxins also might result from breathing air containing trace amounts of dioxins, inadvertently ingesting soil containing dioxins, and absorbing minute levels of dioxins present in the soil through the skin.
The most toxic of the PCDDs is the 2,3,7,8-TCCD congener. Other PCDDs with chlorine in the 2,3,7,8 positions appear to produce similar adverse health effects, but at higher exposure concentrations. When calculating human exposures, dioxins are measured in picograms—that is, trillionths (0.000000000001) of a gram. Highly sophisticated measurement techniques and technologies are required to test for the presence of the 2,3,7,8 substituted dioxin congeners.
Adapted from:
Toxicological Profile for Chlorinated Dibenzo-P-Dioxins. U.S. Department of Health and Human Services, Public Health Service. Agency for Toxic Substances and Disease Registry. December 1998.