Solidification/Stabilization
Application
Many factors figure into whether S/S can be a viable alternative for treatment at a site. The following questions (and drop-down discussions) provide an initial screening process when considering S/S as a remedy:
S/S has been tested and evaluated for its effectiveness in immobilizing a wide array of contaminants such as metals, creosote, and petroleum products. According to EPA (1993 and 2000) and ITRC (2011), S/S has been demonstrated1 for treating the following COCs:
Organic COCs
- Semi-volatile organic compounds (SVOCs)
- Non-volatile organic compounds
- Polychlorinated biphenyls (PCBs)
- Pesticides
- Dioxins/furans
- Halogenated and non-halogenated volatile organic compounds (VOCs), with pretreatment
Inorganic COCs
- Volatile and non-volatile metals
- Asbestos
- Radioactive materials
- Inorganic corrosives
- Inorganic cyanides
Reactive COCs
- Oxidizers (e.g., sodium hypochlorite, potassium permanganate, nitric acid, or potassium dichromate) (EPA, 1993)
As an initial screening consideration, the presence of most COCs does not rule out S/S as an alternative at a site, although some COCs require additives for successful immobilization. For example, although most metals form insoluble compounds when combined with cementitious binders, arsenic(III), chromium(VI), and mercury require pretreatment or the use of additives such as lime (Mulligan et al, 2001, cited in Paria and Yuet, 2006). In addition, treatment of metals with cementitious binders becomes less effective with increasing concentrations of organic compounds (ITRC, 2011) because hydrophobic organic compounds can interfere with the hydration reactions of cement or pozzolanic materials and inhibit setting (Paria and Yuet 2006). However, use of additives can increase binder effectiveness and immobilize organics.
As discussed later in this section, treatability testing can help evaluate performance of an S/S remedy using site-specific contaminated material.
As with other remedies, the size and depth of the contaminated area will affect the time and cost to implement S/S. Larger contaminated areas require more treatment reagents and mixing, and subsurface variability is more likely. As a result, larger areas of contamination might require different proportions of reagents or mixing approaches. Limiting factors for the depth of the contaminated material include:
- Ex situ S/S is better suited to smaller, shallow source zones above the water table that can be reached by conventional excavation equipment (maximum 20 feet below ground surface).
- In situ S/S can achieve greater treatment depths (up to 60-100 feet below ground surface) using augers to inject and mix reagents (EPA, 1997; ITRC, 2011).
- In situ S/S design in northern climates must consider the effects of deep freezing, permafrost, spring melt and frost heave (Government of Canada, 2017).
- Dewatering may be necessary for in situ mixing within the saturated zone (Government of Canada, 2017).
Physical aspects of the site can limit the feasibility of implementing S/S. Logistical considerations include:
- Remote locations might be prohibitively difficult to access for delivering and operating equipment and materials (e.g., poor roads, limited access to water or power).
- Site topography (e.g., steep hills and uneven surfaces) might limit equipment access to contaminated areas.
- The presence of overhead utility lines and trees might limit equipment access and operation, particularly where large excavators or drilling rigs are needed.
- Underground utilities, buried debris, and other obstacles within the contaminated area will interfere with effective mixing and must be removed.
- Adequate space is needed to accommodate storage of excavation and mixing equipment as well as stockpiles of reagents and water.
- The site should be accessible for required long-term monitoring.
Site geology will strongly influence adequate delivery and thorough mixing of reagents with the contaminated media. Some geological considerations for S/S applications include:
- S/S is best suited for unconsolidated soil and sediment deposits or sludge and is not applicable in bedrock environments.
- Implementation is generally easier in sandy, silty, or gravelly soils.
- Cobbles and boulders need to be removed. Excessive quantities of coarse particles may inhibit the effectiveness of S/S remedies (Bates and Hills, 2015).
- Specialized mixing may be needed to treat clayey soils to prevent clay balls of unmixed and untreated material (Ricciardi-Rigault et al., 2017).
- The presence of organic matter might affect the effectiveness of some binders or require the use of additives.
- Sites in seismically active zones may not be appropriate for placement of S/S-treated material.
- Subsurface heterogeneity can affect in situ mixing and treatment, which can result in uneven performance.
Prior to implementing a remedy, remedial action objectives (RAOs) are established to describe what the remedial action needs to accomplish. RAOs generally identify the COCs, environmental media, exposure pathways, receptors to be protected, and the cleanup levels that must be achieved based on current and future land use. To get a sense of whether S/S can meet RAOs at a specific site, case studies and other resources describing similar full-scale applications of S/S can be reviewed. The Examples of Site-Specific Use section lists case studies and other resources that may be useful. Consider also that incorporation of other remedy components can help S/S meet RAOs. These components might include containment technologies (e.g., a cap or slurry walls to limit infiltration of water) or a downgradient groundwater remedy to address residual groundwater contamination not treated by S/S.
If the answers to questions 1-5 indicate that S/S could be a viable remedial alternative, a general process is followed that typically includes:
- Establishing material performance specifications based on remediation goals.
- Treatability studies to design an appropriate mix of reagents and additives that effectively meet material performance specifications and refine implementation techniques and construction performance specifications.
- Mobilization of field equipment to implement the S/S mix design at field scale.
- Monitoring of material performance after the remediation process is completed (ITRC, 2011).
Material performance specifications are design targets developed based on the material performance goals and used during treatability studies to evaluate whether a treated material will meet the goals established at the start of the S/S design and implementation process (ITRC, 2011). The most common performance parameters used in material performance specifications to assess the S/S-treated product include: unconfined compressive strength, hydraulic conductivity (to determine water entry into the S/S product), and leachability.
Treatability tests are typically bench-scale investigations that characterize the untreated contaminated material (e.g., the array of contaminants and concentrations and the presence of constituents or materials that might interfere with efficacy of binders) and evaluate performance of the S/S remedy under different operating conditions reflecting current and anticipated site conditions (e.g., climate and site reuse). Multiple formulations of binders and additives may be tested. Results are used to develop process design parameters (e.g., types of reagents and mixing approach) and scale up for full-scale implementation to treat the full extent of contaminated material. Because of the variable nature of contaminated materials (particularly soil), bench-scale treatability testing to evaluate the effectiveness of potential binders and additives is a prerequisite to testing S/S in the field.
Although not a substitute for treatability testing and pilot tests, the following questions and answers summarize options for binders, additives, and mixing, as well as some of the tests available to evaluate the established performance specifications.
The selection of binders and additives for use in S/S must address: (1) compatibility between the reagents and the materials being treated, including any unforeseen interactions; (2) the presence of chemicals that interfere with the setting and durability of the product; and (3) anticipated ground and groundwater conditions over the long term. The nature of contaminants can vary across a site requiring remediation, which means that more than one binder formulation might be required.
Portland cement and pozzolans, or a mixture of the two, are the most commonly used binders for S/S. Pozzolans are silica- or alumina-rich inorganic materials (commonly process wastes, such as fly ash and cement kiln dust) that can react with calcium hydroxide to form compounds with cement-like properties. These binders are effective for a range of inorganic cations and anions.
Used less frequently than Portland cement and other cementitious binders, polymer binders are thermoplastic or thermosetting materials that are poured as a liquid over and around the contaminated material to enclose it, forming a solidified block when cooled (EPA, 1997). While effective, polymer binders are usually employed ex situ and can be difficult to use in an in situ setting (EPA 2006). Thermoplastic binders (e.g., polyethylene, sulfur polymer, and bitumen) can be melted repeatedly to a flow state and harden when cooled. Thermosetting binders require the combination of several liquid ingredients (e.g., monomer, catalyst, and promoter) that, when combined, harden to a solid that cannot be reworked (EPA 1997). Thermoplastic binders operate in a temperature range of 120 to 180°C, which could be an issue in soil with high moisture content. Thermosetting binders operate at ambient temperatures, but they too are not amenable to high moisture content.
Additives either target specific contaminants to further reduce their leachability or improve the cure/set time and other properties of the treated material. Additives may be incorporated in the waste first as a pretreatment or mixed in the waste concurrently with the binder.
Sorbents are additives that have adsorptive properties that transfer and bind organic contaminants to the surface of the sorbent to reduce their mobility. For example, when cement or pozzolans are used as a binder in S/S, most organic compounds are physically entrapped within the treated material rather than chemically changed; incorporating a sorbent adds another mechanism to reduce their leachability. Examples of sorbents used in S/S include organophilic clays, bentonite, activated carbon phosphates, rubber particulates (e.g., from shredded tires), and chemical gellants (ITRC, 2011).
Phosphates are additives primarily used to stabilize lead and other heavy metals contamination. They chemically react with metals contamination to form low-solubility compounds or metal complexes (molecules with a central metal cation surrounded by other ions or molecules). Their low solubility decreases the mobility of the metals. Examples of phosphate additives include trisodium phosphate and the mineral apatite. Phosphates also can be added to cementitious materials to improve the physical characteristics of the treated waste or as a pretreatment step to a solidification process.
Lime additives such as quicklime (calcium oxide) and hydrated lime (calcium hydroxide) can be mixed with contaminated clayey soil to raise soil pH to around 12.5. This promotes the dissolution of the silica and alumina clay structure, and the result is a tough, water-insoluble gel that cements the soil particles (Bone et al. 2004); however, cyclic wetting and drying of lime-stabilized soil can break down the cementitious bonds. The addition of lime reduces the plasticity and increases the shear strength of soils, which facilitates soil handling.
Lime can also be used with some fly ashes to form compounds that possess cementitious properties. The stabilization effect of fly ash relies on the formation of calcium silicate gels, which gradually harden over time to form a stable material. Fly ash-lime products containing waste materials possess favorable leaching characteristics, especially for waste containing heavy metals, where the metal ions may be chemically bound to the hydrate complexes (Al-Tabbaa and Perera 2002).
Reagents require thorough mixing with the contaminated material to form a homogenous mixture that can meet performance parameters. An S/S treated material must withstand external forces (e.g., freeze-thaw cycles, seismic activity, and precipitation) that could increase contaminant leaching and mobility. Underground obstructions, such as buried debris, are first removed to improve mixing performance. Samples of the mixture may be collected systematically during the mixing process to ensure adequate blending of materials is occurring.
Available mixing options for S/S are dictated by whether it is implemented in situ or ex situ:
For in situ mixing, excavators can be equipped with a mixing head (Figure 1) or with hollow forks or rake attachments through which slurried binder and additives are injected into the ground and mixed by the attachment. The depth of mixing with excavators is limited by the size of the equipment, but generally cannot reach depths greater than 20 feet.
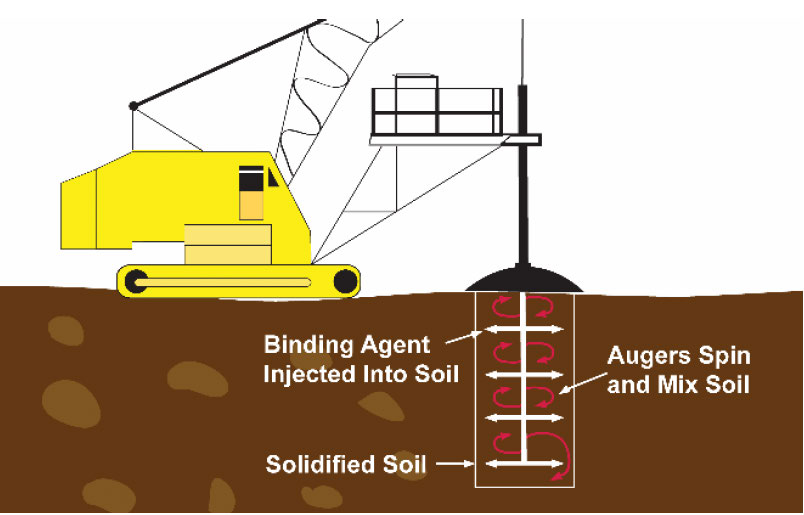
Augers are generally used for deeper injection and mixing (Figure 2) and can reach soils 60 to 100 feet deep (ITRC, 2011; EPA, 1997). Large, flighted rotary augers, that are 6 to 8 feet (or more) in diameter, emplace slurried binders and additives through the auger flights (Figure 2). As the augers bore into the contaminated material, binders are emplaced, creating a large "plug" of treated material. After thorough mixing by the auger rotation, the auger is withdrawn. The position of the auger is advanced to slightly overlap the last plug, and the process is repeated until the contaminated area is mixed (U.S. ACE, 2003).
In situ treated material sets (hardens) in place. The S/S-treated material may increase in volume due to the addition of binders and additives and possible expansion during setting reactions. The degree of expansion and its potential impact on surrounding structures must be considered in the remedy design.
Ex situ mixing is preceded by the excavation and staging the contaminated materials (Figure 3). The materials are typically screened to remove large items (usually 2 inches in diameter or greater) that cannot be treated effectively. Ex situ mixing can be accomplished in pug mills or rotary drum mixers in a fixed or mobile plant.
A significant consideration in applying the S/S ex situ is the "swell factor" in the solid volume created by the binding agent; this factor depends on the amount of reagent added and can approach 50 percent in some cases. Due to the swell factor, the treated material may not fit in the excavation from which it was removed, without altering the natural grade (U.S. ACE, 2003). Al-Tabbaa and Perera (2006) provide a detailed discussion of mixing technologies with accompanying photographs of the equipment used.
Key performance parameters for S/S include compressive strength, hydraulic conductivity, and leachability of the treated product (ITRC, 2011).
Compressive strength of a material relates to its capacity to withstand a load that tends to reduce its size (compression). It is typically measured in S/S-treated materials to ensure that a chemical reaction of binder and water has taken place (ITRC, 2011). Tests are conducted in a laboratory using samples of material prepared during treatability studies and collected during full-scale implementation (e.g., core samples). At a minimum, the S/S material will have at least as much bearing strength as the surrounding soil to support the loads imposed by the equipment used in implementation; however, high compressive strength may be required depending on other considerations, such as future use of the site (ITRC, 2011).
Hydraulic conductivity represents the overall ability of water to flow through the S/S-treated material. The hydraulic conductivity of the treated material, particularly as it compares to surrounding soil, is a measure of water contact. Increased contact with water will increase leachability of contaminants from the treated material. Water will tend to flow around the treated material if it is less conductive than the surrounding soil, but through it if it is more conductive. Hydraulic conductivities similar to silty clay or lower are desirable to minimize the potential for contaminant migration from the treated material (ITRC, 2011).
Leachability is the potential for contaminants to be transferred from the treated material into water that comes into contact with it. There are several tests available to evaluate the leachability of COCs. The final disposal environment intended for the treated waste is an important consideration when determining the appropriate test (Barnett et al. 2009).
Historically, the most commonly specified test has been the Toxicity Characteristic Leaching Procedure or TCLP. The TCLP is linked to regulations in EPA's RCRA program which set the precedent for use of this test in CERCLA. Because this test was designed for evaluation of industrial/municipal landfill leachate, applying the TCLP to S/S-treated waste might not be appropriate when the treated waste is managed other than in a landfill. The TCLP procedure relies on extracting sample waste with a diluted organic acid, thus simulating conditions of mixed-waste disposal (including organic waste), such as that found in a municipal landfill. However, S/S-treated wastes are commonly disposed of in monofils (landfills for disposal of one type of waste) or treated in situ and left in place. Considering this difference, the Synthetic Precipitation Leaching Procedure (SPLP), designed to simulate waste exposure to acid rain, may be more appropriate than the TCLP.
The Leaching Environmental Assessment Framework (LEAF) is a leaching evaluation system, which consists of four leaching methods, data management tools, and scenario assessment approaches (designed to work individually or to be integrated) to describe the release of inorganic constituents of potential concern (COPCs) for a wide range of solid materials. The LEAF Methods have been designed to consider the effect of key environmental conditions and waste properties on contaminant leaching.
- Method 1313. Liquid-Solid Partitioning as a Function of Extract pH Using a Parallel Batch Extraction Procedure.
- Method 1314. Liquid-Solid Partitioning as a Function of Liquid-Solid Ratio for Constituents in Solid Materials Using an Up-Flow Percolation Column Procedure.
- Method 1315. Mass Transfer Rates of Constituents in Monolithic or Compacted Granular Materials Using a Semi-Dynamic Tank Leaching Procedure.
- Method 1316. Liquid-Solid Partitioning as a Function of Liquid-to-Solid Ratio in Solid Materials Using a Parallel Batch Procedure.
References:
Al-Tabbaa, A. and A.S.R. Perera. 2002. ![]() Binders & Technologies, Part I: Basic Principles.
Binders & Technologies, Part I: Basic Principles.
Al-Tabbaa, A. and A.S.R. Perera. 2006. UK Stabilization/Solidification Treatment and Remediation, Part 1: Binders & Technologies.
Barnett, F., S. Lynn, and D. Reisman. 2009. ![]() Technology Performance Review: Selecting and Using Solidification/Stabilization Treatment for Site Remediation. EPA 600-R-09-148.
Technology Performance Review: Selecting and Using Solidification/Stabilization Treatment for Site Remediation. EPA 600-R-09-148.
Bates, E. and C. Hills. 2015. ![]() Stabilization and Solidification of Contaminated Soil and Waste: A Manual of Practice. Hygge Media.
Stabilization and Solidification of Contaminated Soil and Waste: A Manual of Practice. Hygge Media.
Battelle, 2002. ![]() Advances in Encapsulation Technologies for the Management of Mercury-Contaminated Hazardous Wastes. Prepared for U.S EPA National Risk Management Research Laboratory.
Advances in Encapsulation Technologies for the Management of Mercury-Contaminated Hazardous Wastes. Prepared for U.S EPA National Risk Management Research Laboratory.
Bone, B. D. et al. 2004. ![]() Review of Scientific Literature on the Use of Stabilisation/Solidification for the Treatment of Contaminated Soil, Solid Waste, and Sludges. Environment Agency, UK, Science Report SC980003/SR2.
Review of Scientific Literature on the Use of Stabilisation/Solidification for the Treatment of Contaminated Soil, Solid Waste, and Sludges. Environment Agency, UK, Science Report SC980003/SR2.
EPA. 2006. ![]() In Situ Treatment Technologies for Contaminated Soil. EPA 542-F-06-013. Office of Solid Waste and Emergency Response.
In Situ Treatment Technologies for Contaminated Soil. EPA 542-F-06-013. Office of Solid Waste and Emergency Response.
EPA, 2000. ![]() Solidification/Stabilization Use at Superfund Sites. EPA 542-R-00-010. Office of Solid Waste and Emergency Response.
Solidification/Stabilization Use at Superfund Sites. EPA 542-R-00-010. Office of Solid Waste and Emergency Response.
EPA, 1997. ![]() Innovative Site Remediation Design and Application, Volume 4: Stabilization/Solidification. EPA 542-B-97-007. Office of Solid Waste and Emergency Response.
Innovative Site Remediation Design and Application, Volume 4: Stabilization/Solidification. EPA 542-B-97-007. Office of Solid Waste and Emergency Response.
EPA, 1993. ![]() Engineering Bulletin: Solidification/Stabilization of Organics and Inorganics. EPA 540-S-92-015. Office of Research and Development.
Engineering Bulletin: Solidification/Stabilization of Organics and Inorganics. EPA 540-S-92-015. Office of Research and Development.
Government of Canada, 2017. Factsheet: Solidification/Stabilization–In Situ. –Ex Situ.
ITRC (Interstate Technology & Regulatory Council), 2011. ![]() Development of Performance Specifications for Solidification/Stabilization.
Development of Performance Specifications for Solidification/Stabilization.
Paria, S. and P.K. Yuet. 2006. ![]() Solidification/Stabilization of Organic and Inorganic Contaminants Using Portland Cement: A Literature Review. Environmental Reviews 14(4):217-255.
Solidification/Stabilization of Organic and Inorganic Contaminants Using Portland Cement: A Literature Review. Environmental Reviews 14(4):217-255.
Ricciardi-Rigault et al., 2017. Guidance and Orientation for the Selection of Technologies – Contaminated Sites – Pollution and Waste – Environmental and Natural Resources, Canada.ca
Sorengard, M., et al., 2021. Laboratory-Scale and Pilot-Scale Stabilization and Solidification (S/S) Remediation of Soil Contaminated with Per- and Polyfluoralkyl Substances (PFASs). Journal of Hazardous Materials 402:123453 (Abstract)
Sorengard, M., et al., 2019. Stabilization and Solidification Remediation of Soil Contaminated with Poly- and Perfluoroalkyl Substances (PFASs). Journal of Hazardous Materials 367:639-646 (Abstract)
U.S. ACE (U.S. Army Corps of Engineers), 2003. ![]() Safety and Health Aspects of HTRW Remediation Technologies. EM 1110-1-4007, p 4-1 - 4-12.
Safety and Health Aspects of HTRW Remediation Technologies. EM 1110-1-4007, p 4-1 - 4-12.
U.S. ACE (U.S. Army Corps of Engineers), 1995. ![]() Engineering and Design Treatability Studies for Solidification/Stabilization of Contaminated Material. ETL 1110-1-158.
Engineering and Design Treatability Studies for Solidification/Stabilization of Contaminated Material. ETL 1110-1-158.
Helpful Information
Note that S/S of emerging contaminants such as per- and polyfluoroalkyl substances (PFAS), is in the research stage and has not been demonstrated, but recent research indicates that S/S may be applicable for soil remediation (Sorengard, et al., 2021 and 2019). ↩
Note that S/S of emerging contaminants such as per- and polyfluoroalkyl substances (PFAS), is in the research stage and has not been demonstrated, but recent research indicates that S/S may be applicable for soil remediation (Sorengard, et al., 2021 and 2019). ↩